Acidic Names.
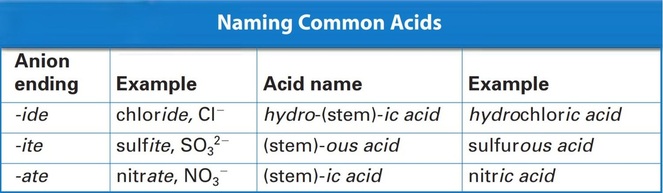
Acids are Ionic Compounds however, a special group of them. Acids could be simply defined as ionic compounds with one or more Hydrogen Atoms that produces Hydrogen ions. Acid Formulas are basically an Anion combined with the amount of Hydrogen atoms necessary to make the acid electrically neutral. Usually it is written as HnX with n equalling the amount of Hydrogens to in fact make the acid neutral. The table above explains how to name acids. Using the anion ending, you can find the name for the acid using the endings. If the anion therefore ends in -ide you start with hydro use the stem of the anion, add -ic and finish with acid. For the anion ending -ite add -ous acid to the stem. And for -ate anion endings, add -ic acid to the stem of the anion.
