Naming Molecules
Non-metals are any element to the right side of the steps of the periodic table. For example Hydrogen, Helium, Oxygen, and Nitrogen are just a few non-metals. When two or more non-metals combine they combine covalently. Covalents bonds occur when two or more non-metals share electrons to fill their outter valence shell. The element with the lesser electronegativity acts as the metal and the one with the higher electronegativity pulls the electrons closer to it.
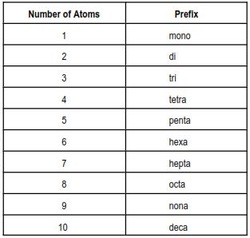
To name these compounds, there are two parts in the name. Both use prefixes located in the table to the left. The first part of the compound is named with the full element name of the element with the lower electronegativity this will have a prefix before it, unless there is only one in which you do not need the mono- prefix. For the second part of the name, take the quantity of the element in the compound and add that prefix. As for its suffix, add -ide to the end.
Prefix #1 + name of first element + Prefix #2 + root of second element + ide.
Examples: CO - (only one Carbon, no prefix needed)+Carbon +Mon(o)+Ox + ide. = Carbon Monoxide.
H20 - Di + Hydrogen + Mon(o) + Ox + ide. = Dihydrogen Monoxide.
N2O5 - Di + Nitrogen + Pent(a) + Ox + ide. = Dinitrogen Pentoxide.
To name these compounds, there are two parts in the name. Both use prefixes located in the table to the left. The first part of the compound is named with the full element name of the element with the lower electronegativity this will have a prefix before it, unless there is only one in which you do not need the mono- prefix. For the second part of the name, take the quantity of the element in the compound and add that prefix. As for its suffix, add -ide to the end.
Prefix #1 + name of first element + Prefix #2 + root of second element + ide.
Examples: CO - (only one Carbon, no prefix needed)+Carbon +Mon(o)+Ox + ide. = Carbon Monoxide.
H20 - Di + Hydrogen + Mon(o) + Ox + ide. = Dihydrogen Monoxide.
N2O5 - Di + Nitrogen + Pent(a) + Ox + ide. = Dinitrogen Pentoxide.
