Common Polyatomic Ions Table
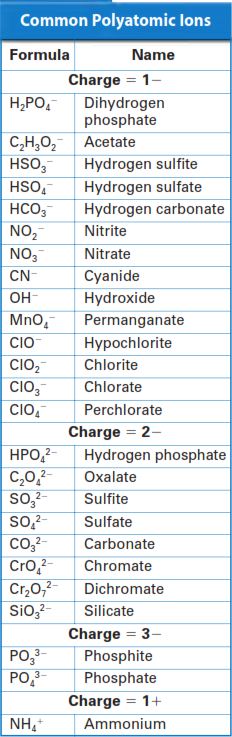
The table to the left here is a table of the most common Polyatomic Ions that you would see in most frequently while dealing with Polyatomic Ions. Before we discuss this, let's discuss the term Polyatomic. Poly- the prefix means more than one, (Mono would be one). For example, Polyphony in music theory is many sounds. Atomic refers to atoms, like Hydrogen atoms or Carbon atoms. Therefore putting these together means more than one atom, Polyatomic Ions, more than one atom in the Ions. Ionic Compounds almost.
Most commonly, Polyatomic anions will have the endings of -ite or -ate. For example, NO2- is Nitrite and NO3- is Nitrate. Additionally notice though, that the positively charged NH4+ ends in -ium, and both CN- and OH- end in -ide. (NH4+ is Ammonium, CN- is Cyanide, and OH- is Hydroxide)
For most of the ions you just have to memorize the name for them. However some patterns are in them. An Anion is an ion with a negative charge. A Cation is an ion with a positive charge. So all of the polyatomic ions listed besides Ammonium (NH4+) are Anions. Ammonium is therefore a Cation.
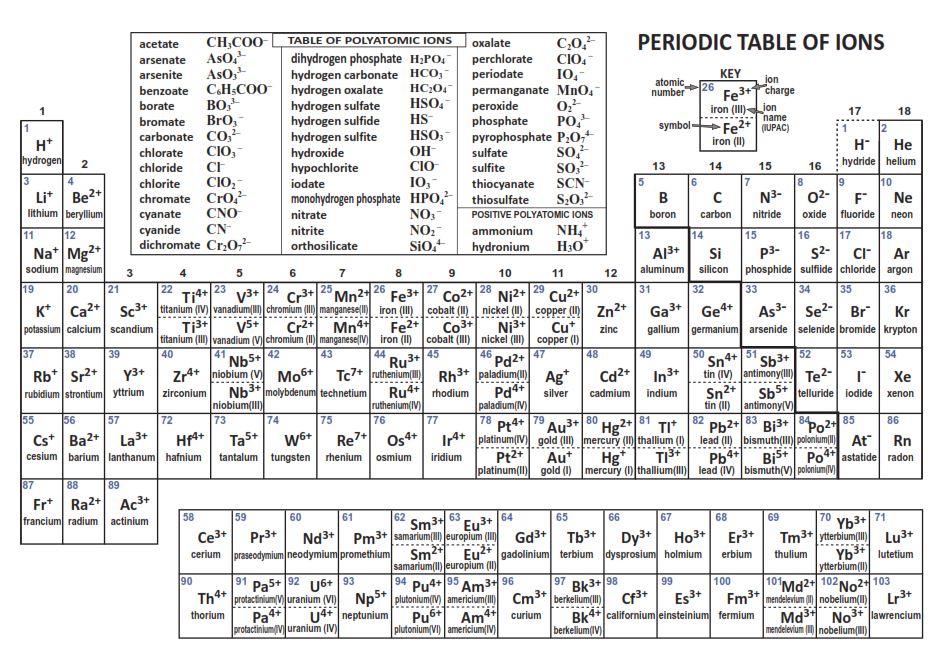
Remember however, when naming all of ionic compounds, any of the ions below that have different charges must be taken into consideration when naming them. For example, when using Fe2+ use Iron (II), when using Fe3+ use Iron (III) therefore when you name them include the roman numeral in the compound. There is a periodic table below to decide which ions have multiple charges and which to use.
Most commonly, Polyatomic anions will have the endings of -ite or -ate. For example, NO2- is Nitrite and NO3- is Nitrate. Additionally notice though, that the positively charged NH4+ ends in -ium, and both CN- and OH- end in -ide. (NH4+ is Ammonium, CN- is Cyanide, and OH- is Hydroxide)
For most of the ions you just have to memorize the name for them. However some patterns are in them. An Anion is an ion with a negative charge. A Cation is an ion with a positive charge. So all of the polyatomic ions listed besides Ammonium (NH4+) are Anions. Ammonium is therefore a Cation.
Remember however, when naming all of ionic compounds, any of the ions below that have different charges must be taken into consideration when naming them. For example, when using Fe2+ use Iron (II), when using Fe3+ use Iron (III) therefore when you name them include the roman numeral in the compound. There is a periodic table below to decide which ions have multiple charges and which to use.